Advantages
- Demonstrated recovery of Co, Ni, and Mn in mixtures of each
- Enables quick (10 min) and highly selective (>90%) recovery
- Recovery by 14-membered ring azamacrocyclic ligand (H2HAM)
- Low environmental impact / simple handling / no need for specialized equipment, allowing for processability at any scale
Background & Technology
Lithium ion batteries (LIBs) contain a number of valuable metals such as nickel, cobalt, and manganese. The metal content of waste LIB is much higher than that in ore, and its recovery has attracted much attention. In metal recovery from ternary lithium-ion batteries, many studies have applied hydrometallurgical methods, such as eluting metal ions into solutions and subsequent separation and recovery. However, leachate from waste ternary lithium-ion batteries contain many types of metals, and it is difficult to separate and regenerate a single metal ion from such solutions in an efficient and environmentally friendly manner, and no method has been established to recover each single metal.
This technology is a method for selectively separating and recovering metal ions from mixed solutions of multiple metal ions using H2HAM to solve these problems. H2HAM is a compound characterized by relatively small N4 space and forms different products depending on the ionic radii of Co(II), Ni(II), and Mn(II) ions. For example, the reaction with Co(II) ions produces a CoN4 complex with an octahedral structure, the reaction with Ni(II) ions produces a planar tetracoordinated NiN4 complex, and the reaction with Mn(II) ions produces a Mn(N4)2 complex with a sandwich structure. Since the solubility of these products is low, all products can be recovered in high yield as precipitation. Furthermore, the inventor found that there is a pecking order of Co > Ni > Mn in the precipitation rate. Using these phenomena, Co, Ni, and Mn are precipitated in this order, and each can be easily recovered as a precipitate while being separated.
 |
Data
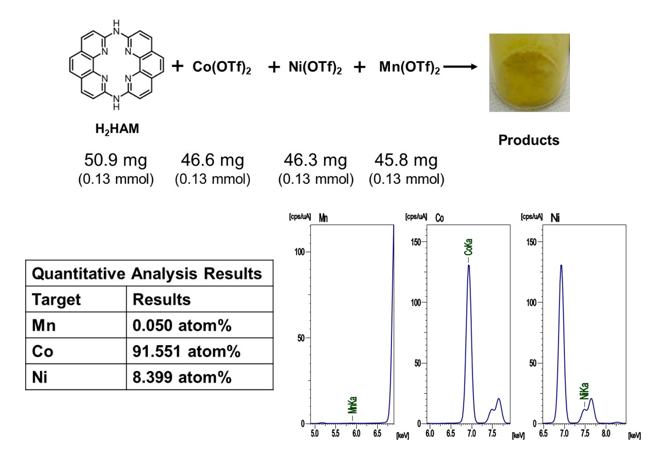
Competitive tests in the coexistence of Co, Ni, and Mn ions
H2HAM, Co(OTf)2, Ni(OTf)2, and Mn(OTf)2 at a molar ratio of 1:1:1 resulted in the preferential formation of Co complexes, and H2HAM recognized Co among Co, Ni and Mn.
 |
Patent
Pending
Researcher
Shizuoka University Graduate School of Integrated Science and Technology Department of Chemistry Course Associate Professor Makoto Moriya
Development Phase
Current Stage: Proof of concept for single recovery of multiple metal ions with H2HAM is complete.
Next stage:
1) Demonstration and development of recovery and recycling processes for ligands (in progress)
2) Development of efficient processes for recovery of each metal
3) Exploration of applications and process demonstration for other purposes
We are looking for partner companies interested in introducing this technology or collaborating in its practical application / development. We also welcome proposals from companies regarding 3).
We would be happy to start with a detailed explanation and discussion of the technology.
Project No.ON-05234


