Advantages
- Low-cost and high-safety because the magnetic field is extremely weak
- New treatments for psychiatric and neurological disorders such as Parkinson’s disease and depression
Background
We have identified that mitochondrial phagocytosis (mitophagy) is activated and activates mitochondria by physiological magnetic stimulation by extremely low frequency fluctuation of weak magnetic fields (ELF-WMF). Furthermore, we developed a new method of ELF-WMF with a specific pattern that more effectively activates cell mitophagy, and confirmed the therapeutic effect on Parkinson’s disease and depression animal models. We have identified a specific molecule on which ELF-WMF acts, and are currently studying the mechanism leading to mitophagy activation.
Data
- In each cell of HEK (human kidney cell), AML12 (mouse hepatocyte), C2C12 (mouse striated muscle cell), Neuro2a (mouse neuroblast), mitochondria were decreased by exposure to this weak magnetic field.
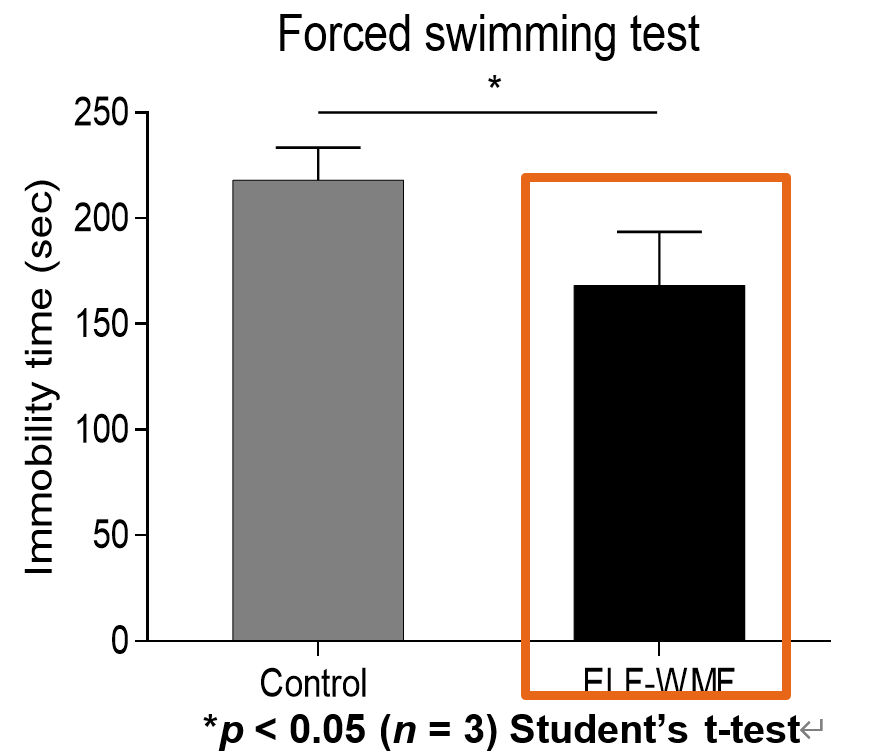
- Exposure to Parkinson’s disease model ASO mice improves athletic performance (upper figure)
- Depression symptoms improved by exposure to depression model mice (lower figure)
 |
 |
Patent
Patent applied (unpublished)
Researcher
Kinji Ohno, Professor, Graduate School of Medicine Center for Neural Disease and Cancer, Nagoya University
Goals
- POC in animal study has been completed by using our prototype device at the current stage.
- We are looking for collaborative partner companies for practical development, and what we expect from partner companies is to develop a more complete weak magnetic field treatment device. In the future, the goal is to clinically apply it as a therapeutic device after confirming its efficacy and safety in animal experiments.
Product No. BK-03454


