Advantage and Core Benefit
- Natural proteins are easily functionalized.
- Homogeneous proteins site-specifically liked with a molecule are obtained without any reaction on sidechain.
- No limitation of N-terminal amino acid residue.
Potential Applications
- Pharmaceuticals: Antibody-drug conjugates (ADCs), PEGylation, New modalities (e.g. DNA aptamer-antibody Fc conjugates), TR-FRET libraries for drug discovery, Radio-active labeling for pharmacokinetics
- Diagnostics: ELISA/RIA, PET/SPECT
Background and Technology
There are several protein linking methods for functionalization. Some methods non-specifically modify functional groups of protein, which might affect original function of the protein, whereas others need protein modification at a gene design level.
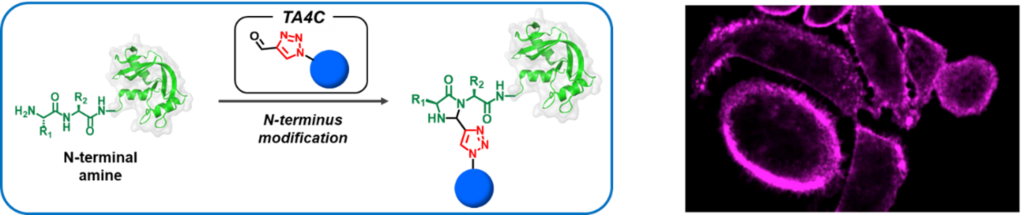
Newly developed linker 1H-1,2,3-triazole-4-carbaldehyde (TA4C) derivatives selectively react with N-terminal amine of proteins, which enable to efficiently introduce new functional groups without modifying original function/structure of protein (fig.L).
 |
Data
- TA4C-payloads are easily synthesized with 1-3 steps using previously reported methods.
- Half-life can be controlled from several hours to one week.
- Fluorescent dye-conjugated trastuzumab shows the same affinity as the original one and fluoresced on the Her2 expressing cell surface (fig.R).
Patent/Publication
- PCT/JP2020/008357; US, EP, CN, JP
- https://doi.org/10.1002/cbic.201900692
Researchers
Prof. Akira Onoda (Hokkaido University)
Expectations
- We are seeking companies to license and commercialize this technology.
- Samples for your evaluation are available under material transfer agreement (MTA).
Project No: KJ-02501


