Advantages
- Screening of patients at high risk of developing interstitial pneumonia
- Detected by ELISA from blood samples
Background
Currently, immune checkpoint inhibitors (ICBs) like PD-1 inhibitors are used to treat various types of cancer, but their side effects, immune-related adverse events (irAEs), have become a serious problem. Ideally, ICB should activate only the anti-tumor immune response, attacking cancer cells while leaving normal cells unaffected. However, ICB sometimes activates the immune response against normal cells (autoimmune response), which is the cause of irAE. Based on prognostic studies of patients treated with PD-1 inhibitors, we revealed that interstitial pneumonia, one of irAEs, was frequently observed in patients with specific HLA types, and comprehensive analysis for 23,000 antigens presented by the patients’ HLA identified specific antigens that induced interstitial pneumonia. Antibody responses against the antigens became biomarkers for risk determination.
Data
 |
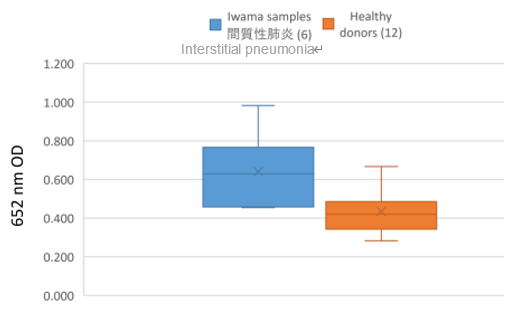 |
Patent
International Patent Pending
Researcher
Hiroyoshi NISHIKAWA Prof. (Tokai National Higher Education and Research System Nagoya University, Graduate School of Medicine)
Current Stage
- Currently analyzing clinical samples, but need to establish more accurate ELISA
- We are looking for partners to help establish ELISA and develop commercial kit products.
Product No: BK-02998


